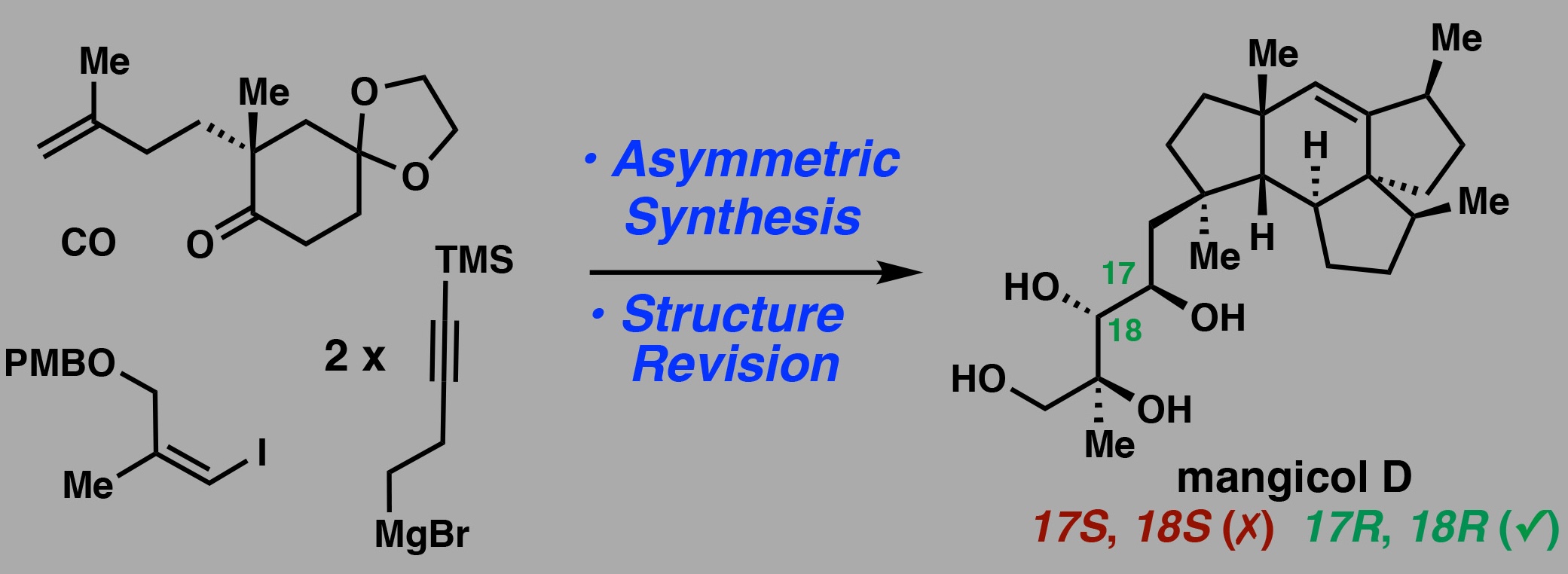
[72] Xie, Y.; Wang, Y.; Zhu, Z.; Ning, S.; Maimone, T.J. Asymmetric Total Synthesis and Structure Revision of (+)-Mangicol D, J. Am. Chem. Soc. 2026, in press.
[71] Stevenson, K.; Moran, D. P.; Maimone, T. J. Catalytic Asymmetric Total Synthesis of Grisemycin, a Thioangucycline Polyketide, J. Am. Chem. Soc. 2026, 148, 11462.

[70] Yu, K.#; Vasilev, V. H.#; Spessert, L. Maimone, T. J. Evolution of a Synthetic Strategy for Complex Diterpenes from Euphorbiaceae and Thymelaeaceae. J. Am. Chem. Soc. 2026, 148, 13102. (# contributed equally)

[69] Ning, S., Maimone, T. J. Total Synthesis of Aconicarmisulfonine A, a Sulfonated Diterpene Alkaloid, J. Am. Chem. Soc. 2025, 147, 46800.

[68] Yu, K.*; Gorou, A.#; Huang, D.#; Maimone, T. J.* Short, Enantioselective Total Synthesis of (+)-Ineleganolide, J. Am. Chem. Soc. 2025, 147, 44727. (# contributed equally) (*co-corresponding authors)

[67] Rosen, H. T.#; Li, K.;# Li, E. L.; Currier, B.; Brittain, S. M.; Garcia, F. J.; Beard, D. C.; Hanna-Holzinger, S.; Dovala, D.; McKenna, J. M.; Schirle, M.; Maimone, T. J.*; Nomura, D. K.* Sulfinyl Aziridines as Stereoselective Covalent Destabilizing Degraders of the Oncogenic Transcription Factor MYC, Angew. Chem. Int. Ed. 2025, 64, e202508518(# contributed equally) (*co-corresponding authors)

[66] Sanchez, A.#; Gonzalez, V. M.#; Sakamoto, J.; Gurajapu, A.; Maimone, T. J. Modular, Enantioselective Entry into Polysubstituted Shapeshifting Molecules, J. Am. Chem. Soc. 2024, 146, 17573. (# contributed equally)

[65] Gowans, F. A.; Thach, D. Q.; Zhu, Z.; Wang, Y.; Altamirano Poblano, B. E.; Dovala, D.; Tallarico, J. A.; McKenna, J. M.; Schirle, M.; Maimone, T. J.*; Nomura, D. K.* Ophiobolin A Covalently Targets Mitochondrial Complex IV Leading to Metabolic Collapse in Cancer Cells, ACS Chem. Biol. 2024, 19, 1260. (*co-corresponding authors)

[64] Harmange Magnani, C. S.; Pinkert, T.; Maimone, T. J. Evaluation of a Cascade Cyclization Approach Toward Harringtonolide, Tetrahedron, 2024, 161, 134072. [Special issue in honor of 2024 Tetrahedron Young Investigator Award]

[63] Harmange Magnani, C. S.; Hernández-Meléndez, J.; Tantillo, D. J.; Maimone, T. J. Total Synthesis of Altemicidin: A Surprise Ending for a Monoterpene Alkaloid, JACS Au 2023, 3, 2883.

[62] Zhu, Z.; Maimone, T. J. Enantioselective Total Synthesis of (–)-Caulamidine A, J. Am. Chem. Soc. 2023, 145, 14215.

[61] Sanchez, A.; Gurajapu, A.; Guo, W.; Kong, W.-Y.; Laconsay, C. J.; Settineri, N. S.; Tantillo, D. J.*; Maimone, T. J.* A Shapeshifting Roadmap for Polycyclic Skeletal Evolution, J. Am. Chem. Soc. 2023, 145, 13452. (*co-corresponding author)

[60] Belcher, B. P.; Machicao, P. A.; Tong, B.; Ho, E.; Friedli, J.; So, B.; Bui, H.; Isobe, Y. Maimone, T. J.*; Nomura, D. K.* Chemoproteomic Profiling Reveals that Anti-Cancer Natural Product Dankastatin B Covalently Targets Mitochondrial VDAC3, Chembiochem, 2023, 24, e202300111. (* co-corresponding authors)

[59] Vasilev, V.; Spessert, L.; Yu, K.; Maimone, T.J. Total Synthesis of Resiniferatoxin, J. Am. Chem. Soc. 2022, 144, 16332.

[58] Xuan, J.; Machicao, P. A.; Haelsig, K. T.; Maimone, T. J. Chemical Investigations of Differentially Oxidized Polycyclic Pyrroles from Bipolaris Fungi: Synthetic Entry Into the Bipolamine Alkaloids, Angew. Chem. Int. Ed. 2022, 61, e202209457.

[57] Sanchez, A.; Maimone, T. J. Taming Shapeshifting Anions: Total Synthesis of Ocellatusone C, J. Am. Chem. Soc. 2022, 144, 7594.

[56] Harmange Magnani, C. S.; Maimone, T. J. Dearomative Synthetic Entry into the Altemicidin Alkaloids, J. Am. Chem. Soc. 2021, 143, 7985.

[55] Tong, B.; Belcher, B. P; Nomura, D. K.; Maimone, T. J. Chemical Investigations Into the Biosynthesis of the Gymnastatin and Dankastatin Alkaloids, Chem. Sci. 2021, 12, 8884.

[54] Reisman, S. E.; Maimone, T. J. Total Synthesis of Complex Natural Products: More Than a Race for Molecular Summits, Acc. Chem. Res. 2021, 54, 1815. (editorial)
[53] Xuan, J.#; Halesig, K. T.#; Sheremet, M.; Machicao, P. A.; Maimone, T. J. Evolution of a Synthetic Strategy for Complex Polypyrrole Alkaloids: Total Syntheses of Curvulamine and Curindolizine, J. Am. Chem. Soc. 2021, 143, 2970. (# contributed equally)

[52] Cho, H.; Shen, Q.; Zhang, L. H.; Okumura, M.; Kawakami, A.; Ambrose, J.; Sigoillot, F.; Miller, H. R.; Gleim, S.; Cobos-Correa, A.; Wang, Y.; Dix, I.; Roma, G.; Eggimann, F.; Moore, C.; Aspesi Jr., P.; Mapa, P. A.; Burks, H.; Ross, N. T.; Krastel, P.; Hild, M.; Maimone, T. J.; Fisher, D. E.; Nomura, D. K.; Tallarico, J. A.; Canham, S. M.; Jenkins, J. L.; Forrester, W. C. CYP27A1 dependent anti-melanoma activity of limonoid natural products targets mitochondrial metabolism, Cell. Chem. Biol. 2021, 28, 1407.

[51] Shen, X.; Thach, D. Q.; Ting, C. P.; Maimone, T. J. Annulative Methods in the Synthesis of Complex Meroterpene Natural Products, Acc. Chem. Res. 2021, 54, 583.
 [50] Luo, M.; Spradlin, J. N.; Boike, L.; Tong, B.; Brittain, S. M.; McKenna, J. M.; Tallarico, J. A.; Shirle, M.; Maimone, T. J.*; Nomura, D. K.* Chemoproteomics-Enabled Discovery of Covalent RNF114-Based Degraders that Mimic Natural Product Function, Cell Chem. Biol. 2021, 28, 559. (*co-corresponding authors)
[50] Luo, M.; Spradlin, J. N.; Boike, L.; Tong, B.; Brittain, S. M.; McKenna, J. M.; Tallarico, J. A.; Shirle, M.; Maimone, T. J.*; Nomura, D. K.* Chemoproteomics-Enabled Discovery of Covalent RNF114-Based Degraders that Mimic Natural Product Function, Cell Chem. Biol. 2021, 28, 559. (*co-corresponding authors)
 [49] Thach, D. Q.; Maimone, T. J. Making Light Work of Lignan Synthesis, Nat. Chem. 2021, 13, 7.
[48] Isobe, Y.; Okumura, M.; McGregor, L. M.; Brittain, S. M.; Jones, M. D.; Liang, X.; White, R.; Forrester, W.; McKenna, J. M.; Tallarico, J. A.; Schirle, M.; Maimone, T. J.*; Nomura, D. K.* Manumycin Polyketides Act as Molecular Glues Between UBR7 and P53, Nat. Chem. Biol. 2020, 16, 1189. (*co-corresponding authors)
[49] Thach, D. Q.; Maimone, T. J. Making Light Work of Lignan Synthesis, Nat. Chem. 2021, 13, 7.
[48] Isobe, Y.; Okumura, M.; McGregor, L. M.; Brittain, S. M.; Jones, M. D.; Liang, X.; White, R.; Forrester, W.; McKenna, J. M.; Tallarico, J. A.; Schirle, M.; Maimone, T. J.*; Nomura, D. K.* Manumycin Polyketides Act as Molecular Glues Between UBR7 and P53, Nat. Chem. Biol. 2020, 16, 1189. (*co-corresponding authors)
 [47] Harmange Magnani, C. S.; Maimone, T. J. Fusicoccin Keeps Getting Stickier: Modulation of an Adapter Protein Interactome with a Molecular Glue Leads to Neurite Outgrowth Cell Chem Biol. 2020, 27, 635-637.
[46] Tong, B.#; Spradlin, J. N.#; Novaes, L. F. T.; Zhang, E.; Hu, X.; Moeller, M.; Brittain, S. M.; McGregor, L. M.; McKenna, J. M.; Tallarico, J. A.; Schirle, M.; Maimone, T. J.*; Nomura, D. K.* A Nimbolide-Based Kinase Degrader Preferentially Degrades Oncogenic BCR-ABL, ACS Chem. Biol. 2020, 15, 1788-1794. (# contributed equally) (*co-corresponding authors)
[47] Harmange Magnani, C. S.; Maimone, T. J. Fusicoccin Keeps Getting Stickier: Modulation of an Adapter Protein Interactome with a Molecular Glue Leads to Neurite Outgrowth Cell Chem Biol. 2020, 27, 635-637.
[46] Tong, B.#; Spradlin, J. N.#; Novaes, L. F. T.; Zhang, E.; Hu, X.; Moeller, M.; Brittain, S. M.; McGregor, L. M.; McKenna, J. M.; Tallarico, J. A.; Schirle, M.; Maimone, T. J.*; Nomura, D. K.* A Nimbolide-Based Kinase Degrader Preferentially Degrades Oncogenic BCR-ABL, ACS Chem. Biol. 2020, 15, 1788-1794. (# contributed equally) (*co-corresponding authors)
 [45] Tong, B.#; Luo, M.#; Xie, Y.; Spradlin, J. N.; Tallarico, J. A.; McKenna, J. M.; Shirle, M.; Maimone, T.J. *; Nomura, D. K.* Bardoxolone Conjugation Enables Targeted Protein Degradation of BRD4, Sci. Rep. 2020, 10, 15543. (# contributed equally) (*co-corresponding authors)
[45] Tong, B.#; Luo, M.#; Xie, Y.; Spradlin, J. N.; Tallarico, J. A.; McKenna, J. M.; Shirle, M.; Maimone, T.J. *; Nomura, D. K.* Bardoxolone Conjugation Enables Targeted Protein Degradation of BRD4, Sci. Rep. 2020, 10, 15543. (# contributed equally) (*co-corresponding authors)
 [44] Harmange Magnani, C. S.; Thach, D. Q.; Haelsig, K. T.; Maimone, T. J. Synthesis of Complex Terpenes from Simple Polyprenyl Precursors, Acc. Chem. Res. 2020, 4, 949-961.
[44] Harmange Magnani, C. S.; Thach, D. Q.; Haelsig, K. T.; Maimone, T. J. Synthesis of Complex Terpenes from Simple Polyprenyl Precursors, Acc. Chem. Res. 2020, 4, 949-961.
 [43] Haelsig, K. T.#; Xuan, J.#; Maimone, T. J. Total Synthesis of (–)-Curvulamine, J. Am. Chem. Soc. 2020, 142, 1206. (# contributed equally)
[43] Haelsig, K. T.#; Xuan, J.#; Maimone, T. J. Total Synthesis of (–)-Curvulamine, J. Am. Chem. Soc. 2020, 142, 1206. (# contributed equally)
 [42] Shen, X.#; Ting, C. P.#; Xu, G.; Maimone, T. J. Programmable Meroterpene Synthesis, Nat. Commun. 2020, 11, 508. (# contributed equally)
[42] Shen, X.#; Ting, C. P.#; Xu, G.; Maimone, T. J. Programmable Meroterpene Synthesis, Nat. Commun. 2020, 11, 508. (# contributed equally)
 [41] Thach, D. Q.#; Brill, Z. G.#; Grover, H. K.; Esguerra, K. V.; Thompson, J. K.; Maimone, T. J. Total Synthesis of (+)-6-epi-Ophiobolin A, Angew. Chem. Int. Ed. 2020, 59, 1532. (# contributed equally)
[41] Thach, D. Q.#; Brill, Z. G.#; Grover, H. K.; Esguerra, K. V.; Thompson, J. K.; Maimone, T. J. Total Synthesis of (+)-6-epi-Ophiobolin A, Angew. Chem. Int. Ed. 2020, 59, 1532. (# contributed equally)  [40] Bersuker, K.; Hendricks, J.; Li, Z.; Magtanong, L.; Ford, B.; Roberts, M. A.; Tong, B.; Maimone, T. J.; Zoncu, R.; Tang, P.; Nomura, D.; Bassik, M. C.; Dixon, S. J.; Olzmann, J. A. The CoQ oxidoreductase FSP1 acts in parallel to GPX4 to inhibit ferroptosis. Nature, 2019, 575, 688.
[39] Hu, X.; Musacchio, A.; Shen, X.; Tao, Y.; Maimone, T. J. Allylative Approaches to the Synthesis of Complex Guaianolide Sesquiterpenes from Apiaceae and Asteraceae. J. Am. Chem. Soc. 2019. 2019, 141, 14904.
[40] Bersuker, K.; Hendricks, J.; Li, Z.; Magtanong, L.; Ford, B.; Roberts, M. A.; Tong, B.; Maimone, T. J.; Zoncu, R.; Tang, P.; Nomura, D.; Bassik, M. C.; Dixon, S. J.; Olzmann, J. A. The CoQ oxidoreductase FSP1 acts in parallel to GPX4 to inhibit ferroptosis. Nature, 2019, 575, 688.
[39] Hu, X.; Musacchio, A.; Shen, X.; Tao, Y.; Maimone, T. J. Allylative Approaches to the Synthesis of Complex Guaianolide Sesquiterpenes from Apiaceae and Asteraceae. J. Am. Chem. Soc. 2019. 2019, 141, 14904.
 [38] Spradlin, J. N.; Hu, X.; Ward, C. C.; Brittain, S. M.; Jones, M. D.; Ou, L.; To, M.; Proudfoot, A.; Ornelas, E.; Woldegiorgis, M.; Olzmann, J. A. Bussiere, D. E.; Thomas, J. R.; Tallarico, J. A.; McKenna, J. M.; Schirle, M.; Maimone, T. J.;* Nomura, D. K.* Harnessing the Anti-Cancer Natural Product Nimbolide for Targeted Protein Degradation, Nat. Chem. Biol. 2019, 15, 747-755 (*co-corresponding authors).
[38] Spradlin, J. N.; Hu, X.; Ward, C. C.; Brittain, S. M.; Jones, M. D.; Ou, L.; To, M.; Proudfoot, A.; Ornelas, E.; Woldegiorgis, M.; Olzmann, J. A. Bussiere, D. E.; Thomas, J. R.; Tallarico, J. A.; McKenna, J. M.; Schirle, M.; Maimone, T. J.;* Nomura, D. K.* Harnessing the Anti-Cancer Natural Product Nimbolide for Targeted Protein Degradation, Nat. Chem. Biol. 2019, 15, 747-755 (*co-corresponding authors).
 [37] Berdan, C. A.; Ho, R.; Lehtola, H. S.; To, M.; Hu, X; Huffman, T. R.; Petri, Y.; Altobelli, C. R.; Demeulenaere, S. G.; Olzmann, J. A.; Maimone, T. J.*; Nomura, D. K.* Parthenolide Covalently Targets and Inhibits Focal Adhesion Kinase in Breast Cancer Cells, Cell Chem Biol. 2019, 26 ,1027. (*co-corresponding author)
[37] Berdan, C. A.; Ho, R.; Lehtola, H. S.; To, M.; Hu, X; Huffman, T. R.; Petri, Y.; Altobelli, C. R.; Demeulenaere, S. G.; Olzmann, J. A.; Maimone, T. J.*; Nomura, D. K.* Parthenolide Covalently Targets and Inhibits Focal Adhesion Kinase in Breast Cancer Cells, Cell Chem Biol. 2019, 26 ,1027. (*co-corresponding author)
 [36] Tong, B.; Maimone, T. J. Enlightening Terpene Synthesis, Chem 2019, 5, 1368-1370
[35] Brill, Z. G.; Zhao, Y.; Vasilev, V.; Maimone, T. J. Synthetic Approaches to the Daphnane and Tigliane Diterpenes from Aromatic Precursors, Tetrahedron, 2019, 75, 4212. [Special issue in honor of Prof. Stephen Buchwald’s Tetrahedron Prize]
[36] Tong, B.; Maimone, T. J. Enlightening Terpene Synthesis, Chem 2019, 5, 1368-1370
[35] Brill, Z. G.; Zhao, Y.; Vasilev, V.; Maimone, T. J. Synthetic Approaches to the Daphnane and Tigliane Diterpenes from Aromatic Precursors, Tetrahedron, 2019, 75, 4212. [Special issue in honor of Prof. Stephen Buchwald’s Tetrahedron Prize]
 [34] Ting, C. P.; Tschanen, E.; Jang, E.; Maimone, T. J. Total Synthesis of Podophyllotoxin and Select Analog Designs via C-H Activation, Tetrahedron, 2019, 75, 3299. [Special issue in honor of Prof. Ryan Shenvi’s Tetrahedron Young Investigator Award]
[34] Ting, C. P.; Tschanen, E.; Jang, E.; Maimone, T. J. Total Synthesis of Podophyllotoxin and Select Analog Designs via C-H Activation, Tetrahedron, 2019, 75, 3299. [Special issue in honor of Prof. Ryan Shenvi’s Tetrahedron Young Investigator Award]
 [33] Hung, K.#; Condakes, M. L.#; Novaes, L. F. T.; Harwood, S. J.; Morikawa, T.; Yang, Z.; Maimone, T. J. Development of a Terpene Feedstock-based Oxidative Synthetic Approach to the Illicium Sesquiterpenes, J. Am. Chem. Soc. 2019, 141, 3083. (# contributed equally)
[33] Hung, K.#; Condakes, M. L.#; Novaes, L. F. T.; Harwood, S. J.; Morikawa, T.; Yang, Z.; Maimone, T. J. Development of a Terpene Feedstock-based Oxidative Synthetic Approach to the Illicium Sesquiterpenes, J. Am. Chem. Soc. 2019, 141, 3083. (# contributed equally)
 [32] Condakes, M. L.; Rosen, R.; Harwood, S. J.; Maimone, T. J. A Copper-catalyzed Double Coupling Enables 3-Step Entry into the Quassinoid Core Architecture, Chem. Sci. 2019, 10,768.
[32] Condakes, M. L.; Rosen, R.; Harwood, S. J.; Maimone, T. J. A Copper-catalyzed Double Coupling Enables 3-Step Entry into the Quassinoid Core Architecture, Chem. Sci. 2019, 10,768.
 [31] Kearney, S. E. et. al. Canvass: A Crowd-Sourced, Natural Product Screening Library for Exploring Biological Space, ACS Cent. Sci. 2018, 4, 1727-1741.
[31] Kearney, S. E. et. al. Canvass: A Crowd-Sourced, Natural Product Screening Library for Exploring Biological Space, ACS Cent. Sci. 2018, 4, 1727-1741.
 [30] Condakes, M. L.; Novaes, L. F. T.; Maimone, T. J. Contemporary Synthetic Strategies Toward Seco-Prezizaane Sesquiterpenes from Illicium Species, J. Org. Chem. 2018, 83, 14843-14852. [JOC Synopsis]
[30] Condakes, M. L.; Novaes, L. F. T.; Maimone, T. J. Contemporary Synthetic Strategies Toward Seco-Prezizaane Sesquiterpenes from Illicium Species, J. Org. Chem. 2018, 83, 14843-14852. [JOC Synopsis]
 [29] Nomura, D. K.;* Maimone, T. J.* “Target Identification of Bioactive Covalently-Acting Natural Products,” in Current Topics in Microbiology and Immunology, Springer, Berlin, Heidelberg, 2018; 1–24. (*co-corresponding author)
[28] Hu, X.; Lim, P.; Fairhurst, R. M.; Maimone, T. J. Synthesis and Study of the Antimalarial Cardamom Peroxide, Tetrahedron, 2018, 74, 3358-3369. [Special issue in honor of Seth Herzon’s Tetrahedron Young Investigator Award]
[29] Nomura, D. K.;* Maimone, T. J.* “Target Identification of Bioactive Covalently-Acting Natural Products,” in Current Topics in Microbiology and Immunology, Springer, Berlin, Heidelberg, 2018; 1–24. (*co-corresponding author)
[28] Hu, X.; Lim, P.; Fairhurst, R. M.; Maimone, T. J. Synthesis and Study of the Antimalarial Cardamom Peroxide, Tetrahedron, 2018, 74, 3358-3369. [Special issue in honor of Seth Herzon’s Tetrahedron Young Investigator Award]
 [27] Hung, K.#; Hu, X.#; Maimone, T. J. Total Synthesis of Complex Terpenoids Employing Radical Cascade Processes, Nat. Prod. Rep. 2018, 35, 174-202. (# contributed equally)
[27] Hung, K.#; Hu, X.#; Maimone, T. J. Total Synthesis of Complex Terpenoids Employing Radical Cascade Processes, Nat. Prod. Rep. 2018, 35, 174-202. (# contributed equally)
 [26] Condakes, M. L.; Hung, K.; Harwood, S. J.; Maimone, T. J. Total Syntheses of (–)-Majucin and (–)-Jiadifenoxolane A, Complex Majucin-Type Illicium Sesquiterpenes, J. Am. Chem. Soc. 2017, 139, 17783-17786.
[26] Condakes, M. L.; Hung, K.; Harwood, S. J.; Maimone, T. J. Total Syntheses of (–)-Majucin and (–)-Jiadifenoxolane A, Complex Majucin-Type Illicium Sesquiterpenes, J. Am. Chem. Soc. 2017, 139, 17783-17786.
 [25] Xu, G.; Elkin, M.; Tantillo, D. J.; Newhouse, T. R.;* Maimone, T. J.* Traversing Biosynthetic Carbocation Landscapes in the Total Synthesis
of Andrastin and Terretonin Meroterpenes, Angew. Chem. Int. Ed. 2017, 56, 12498-12502. (*co-corresponding authors)
[25] Xu, G.; Elkin, M.; Tantillo, D. J.; Newhouse, T. R.;* Maimone, T. J.* Traversing Biosynthetic Carbocation Landscapes in the Total Synthesis
of Andrastin and Terretonin Meroterpenes, Angew. Chem. Int. Ed. 2017, 56, 12498-12502. (*co-corresponding authors)
 [24] Brill, Z. G.; Condakes, M. L.; Ting, C. P.; Maimone, T. J. Navigating the Chiral Pool in the Total Synthesis of Complex Terpene Natural Products, Chem. Rev. 2017, 117, 11753-11795.
[24] Brill, Z. G.; Condakes, M. L.; Ting, C. P.; Maimone, T. J. Navigating the Chiral Pool in the Total Synthesis of Complex Terpene Natural Products, Chem. Rev. 2017, 117, 11753-11795.
 [23] Hu, X.; Xu, S.; Maimone, T. J. A Double Allylation Strategy for Gram-Scale Guaianolide Production: Total Synthesis of (+)-Mikanokryptin, Angew. Chem. Int. Ed. 2017, 56, 1624.
[23] Hu, X.; Xu, S.; Maimone, T. J. A Double Allylation Strategy for Gram-Scale Guaianolide Production: Total Synthesis of (+)-Mikanokryptin, Angew. Chem. Int. Ed. 2017, 56, 1624.
 [22] Hung, K.#; Condakes, M. L.#; Morikawa, T.; Maimone, T. J. Oxidative Entry into the Illicium Sesquiterpenes: Enantiospecific Synthesis of (+)-Pseudoanisatin, J. Am. Chem. Soc. 2016, 138,16616. (# contributed equally)
[22] Hung, K.#; Condakes, M. L.#; Morikawa, T.; Maimone, T. J. Oxidative Entry into the Illicium Sesquiterpenes: Enantiospecific Synthesis of (+)-Pseudoanisatin, J. Am. Chem. Soc. 2016, 138,16616. (# contributed equally)
 [21] Ting, C. P.#; Xu, G.#; Zeng, X.; Maimone, T. J. Annulative Methods Enable a Total Synthesis of the Complex Meroterpene Berkeleyone A, J. Am. Chem. Soc. 2016, 138, 14868. (# contributed equally)
[21] Ting, C. P.#; Xu, G.#; Zeng, X.; Maimone, T. J. Annulative Methods Enable a Total Synthesis of the Complex Meroterpene Berkeleyone A, J. Am. Chem. Soc. 2016, 138, 14868. (# contributed equally)
 [20] Brill, Z. G.; Grover, H. K.; Maimone, T. J. Enantioselective Synthesis of an Ophiobiolin Sesterterpene via a Programmed Radical Cascade, Science 2016, 352, 1078.
[20] Brill, Z. G.; Grover, H. K.; Maimone, T. J. Enantioselective Synthesis of an Ophiobiolin Sesterterpene via a Programmed Radical Cascade, Science 2016, 352, 1078.
 [19] Hu, X., Maimone, T. J. “Peroxy Radical Additions,” in Science of Synthesis, Applications of Domino Transformations in Organic Synthesis, Vol. 1, Snyder, S. A. Ed.; Georg Thieme Verlag: Stuttgart, Germany, 2016; 157-186.
[18] Ting, C.P.; Maimone, T.J. The Total Synthesis of Hyperforin, Synlett 2016, 27, 1443-1449.
[17] Ting, C.P.; Maimone, T. J. Total Synthesis of Hyperforin, J. Am. Chem. Soc. 2015, 137, 10516.
[19] Hu, X., Maimone, T. J. “Peroxy Radical Additions,” in Science of Synthesis, Applications of Domino Transformations in Organic Synthesis, Vol. 1, Snyder, S. A. Ed.; Georg Thieme Verlag: Stuttgart, Germany, 2016; 157-186.
[18] Ting, C.P.; Maimone, T.J. The Total Synthesis of Hyperforin, Synlett 2016, 27, 1443-1449.
[17] Ting, C.P.; Maimone, T. J. Total Synthesis of Hyperforin, J. Am. Chem. Soc. 2015, 137, 10516.
 [16] Zhao, Y.-M.; Maimone, T. J. Short, Enantioselective Total Synthesis of Chatancin, Angew. Chem. Int. Ed. 2015, 54, 1223.
[16] Zhao, Y.-M.; Maimone, T. J. Short, Enantioselective Total Synthesis of Chatancin, Angew. Chem. Int. Ed. 2015, 54, 1223.
 [15] Hu, X.; Maimone, T. J. Four-step Synthesis of the Antimalarial Cardamom Peroxide via an Oxygen Stitching Strategy, J. Am. Chem. Soc. 2014, 136, 5287-5290.
[15] Hu, X.; Maimone, T. J. Four-step Synthesis of the Antimalarial Cardamom Peroxide via an Oxygen Stitching Strategy, J. Am. Chem. Soc. 2014, 136, 5287-5290.
 [14] Ting, C. P.; Maimone, T. J. C-H Bond Arylation in the Synthesis of Aryltetralin Lignans: A Short Total Synthesis of Podophyllotoxin, Angew. Chem. Int. Ed. 2014, 53, 3115-3119.
[14] Ting, C. P.; Maimone, T. J. C-H Bond Arylation in the Synthesis of Aryltetralin Lignans: A Short Total Synthesis of Podophyllotoxin, Angew. Chem. Int. Ed. 2014, 53, 3115-3119.

Before Berkeley
[13] Milner, P. J.; Maimone, T. J.; Su, M.; Chen, J.; Müller, P.; Buchwald, S. L. Investigating the Dearomative Rearrangement of Biaryl Phosphine-Ligated Pd(II) Complexes, J. Am. Chem. Soc. 2012, 134,19922-19934.
[12] Maimone, T. J.; Milner, P. J.; Kinzel, T.; Zhang, Y.; Takase, M. J.; Buchwald, S. L. Evidence for In Situ Catalyst Modification During the Pd-Catalyzed Conversion of Aryl Triflates to Aryl Fluorides, J. Am. Chem. Soc. 2011, 133, 18106-18109.
[11] Maimone, T. J.#; Noël, T.#; Buchwald, S. L. Accelerating Pd-Catalyzed C–F Bond Formation: Use of a Microflow Packed-Bed Reactor, Angew. Chem. Int. Ed. 2011,
50
, 8900-8903. [# contributed equally]
[10] Maimone, T. J.; Buchwald, S. L. Pd-Catalyzed O-Arylation of Ethyl Acetohydroximate: Synthesis of O-Arylhydroxylamines and Substituted Benzofurans, J. Am. Chem. Soc. 2010, 132, 9990-9991.
[9] Maimone, T. J.; Ishihara, Y.; Baran, P. S. Scalable Total Syntheses of (–)-Hapalindole and (+)-Ambiguine H, Tetrahedron 2015, 71, 3652-3665. [Special Issue: The Chemistry of Pyrrole and Indole Natural Products]
[8] Krenske, E. H.; Perry, E. W.; Jerome, S. V.; Maimone, T. J.; Baran, P. S.; Houk, K. N. Why a Proximity-Induced Diels-Alder Reaction Is So Fast, Org. Lett. 2012, 14, 3016-3019.
[7] Maimone, T. J.; Shi, J.; Ashida, S.; Baran, P. S. Total Synthesis of Vinigrol, J. Am. Chem. Soc. 2009, 47, 17066-17067.
[6] Maimone, T. J.; Voica, A. F.; Baran, P. S. A Concise Approach to Vinigrol, Angew. Chem Int. Ed. 2008, 47, 3054-3056.
[5] Richter, J. M.; Whitefield, B. W.; Maimone, T. J.; Lin, D. W.; Castroviejo, M. P.; Baran, P. S. Scope and Mechanism of Direct Indole and Pyrrole Couplings Adjacent to Carbonyl Compounds: Total Synthesis of Acremoauxin A and Oxazinin 3, J. Am. Chem. Soc. 2007, 129, 12857-12869.
[4] Baran, P. S.; Maimone, T. J. A Tuxedo for Iodine Atoms, Nature 2007, 445, 826-827.
[3] Maimone, T. J.; Baran, P. S. Modern Synthetic Efforts Toward Biologically Active Terpenes, Nat. Chem. Biol. 2007, 3, 396-407.
[2] Baran, P. S.; Maimone, T. J.; Richter, J. M. Total Synthesis of Marine Natural Products Without Using Protecting Groups, Nature, 2007, 446, 404-408.
[1] Malerich, J. P.; Maimone, T. J.; Elliot, G. M.; Trauner, D. Biomimetic Synthesis of Antimalarial Naphthoquinones, J. Am. Chem. Soc. 2005, 127, 6276-6283.


















































